

Metal powders are the main functional part in electrode paste, and they also provide conductivity. The amount of metal powder used is important because it greatly affects how well the electrode performs. When heated to high temperatures, the metal particles join together and form a tight, connected network. This structure allows electricity to flow.
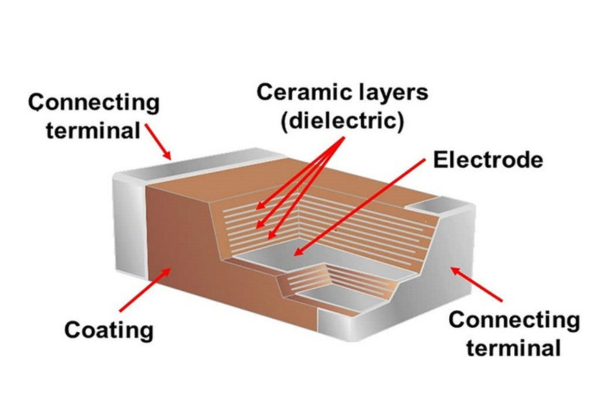
Fig 1. MLCC-and-Ceramic-Capacitors, source: DOEEET
MLCC internal electrodes are usually made from metal powders with high melting points. Examples include palladium-silver alloy, palladium, and nickel. These metals must be fired together with ceramic dielectrics (based on BaTiO₃). The firing happens at high temperatures, usually between 950°C and 1300°C. Because of this, the metals need high melting points and must also be chemically stable. This ensures they do not melt, undergo excessive oxidation, or adversely react with the dielectric at sintering temperatures. Nickel has become the mainstream material for internal electrodes, because it has low cost and low electromigration. Additionally, it shows good co-sintering compatibility with ceramics.
MLCC external electrodes are mainly used to connect the internal electrodes. They typically use metal powder materials like silver and copper. The external electrodes are applied to ceramic bodies that have already been sintered, and then undergo a secondary sintering process. The temperature for this sintering is usually below 900°C, which is much lower than the co-sintering temperature of the internal electrodes and ceramic dielectrics. Therefore, metals with lower sintering temperatures, such as silver and copper, are suitable choices.
MLCC Electrode Paste & Metal Powders Used
|
Electrode Paste Type |
Metal Powders Used |
Powder Requirements |
|
Internal Electrode Paste |
Fine particle size, spherical particles, uniform distribution, and specific surface area |
|
|
Palladium-Silver Alloy (AgPd) |
||
|
Palladium (Pd) |
||
|
External Electrode Paste |
Fine particle size, moderate flakiness, uniform distribution, and specific surface area |
|
Silver and palladium offer excellent conductivity but come at a higher cost. As a result, nickel and copper have been introduced into electrode paste production.
Nickel powder has several key characteristics. It is low in cost and high in electrical conductivity. It demonstrates low electromigration and offers strong corrosion resistance and good heat resistance. It also has a relatively high sintering temperature. Additionally, it works well when co-sintered with ceramic dielectric materials at high temperatures. From a cost standpoint, nickel is much cheaper than precious metals like palladium-silver alloy and pure palladium.
Copper powder also has important advantages. It provides good conductivity and low electrochemical migration. It is also low in cost. The sintering temperature of copper is lower than that of internal electrode materials and ceramic dielectrics. Because of this, copper electrode paste is well-suited for the secondary sintering process that is used to form MLCC external electrodes.
Ultrafine copper powder is an ideal material for making MLCC electrodes. This powder must have high purity, good dispersibility, and consistently uniform small particle size.
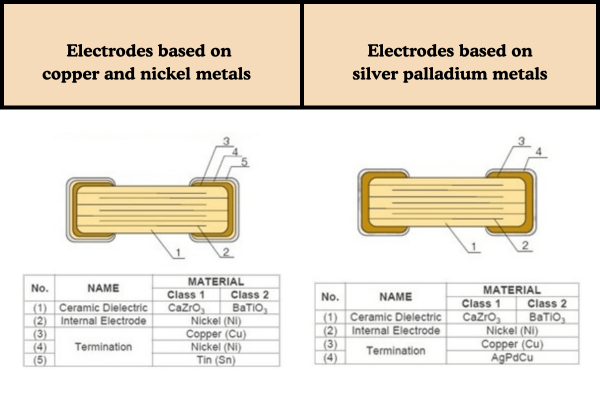
Fig 2. Electrodes based on different metals, source: TDK
Metal powders are the core functional part of MLCC electrode paste, so they must meet a number of strict performance requirements.
In summary, the performance and cost of MLCC electrodes are dictated by their metal powders. Nickel and copper are prevalent due to their optimal balance of conductivity, sintering compatibility, and cost-effectiveness, while precious metals like silver and palladium are used for specialized needs.

United States




