

Cobalt aluminate (CoAl₂O₄) is an inorganic compound with a spinel structure. Industrially, it's mainly used as a blue pigment. You'll find it in ceramics, glass, coatings, and plastics. Lately, it's also been making its way into advanced optical materials.

In the ceramics world, cobalt aluminate is a go-to for underglaze and overglaze decoration. When you add it to the clay body or glaze and fire it above 1200°C, you get a stable, vibrant blue. This blue holds up well—it won't change color due to chemical attack from the molten glaze, and it won't bleed into the surrounding glaze. In enamelware, it's used in the base coat to improve hiding power.
When glass is being made, cobalt aluminate can be added as a colorant. The cobalt ions dissolve in the molten glass, giving it a blue color. This blue glass is used in things like filters, glassware, and decorative items. In cloisonné and other enamel-on-metal crafts, it's mixed with other inorganic pigments to create blue enamel. After firing, the surface is smooth with even color.
Because it holds up so well against weather and chemicals, cobalt aluminate is a great option for outdoor paints and tough engineering plastics. In coil and powder coatings, it can handle the heat of the curing process without any issues, and its color barely changes even after being in the sun for a long time. For plastics, it's perfect for materials that need high processing temperatures, like polyolefins. The end result? Parts that keep their true color, whether they're injection molded or extruded.
Cobalt aluminate has high reflectivity in the near-infrared range. That's why you'll find it in things like energy-efficient building paints. Because the coating reflects infrared heat instead of absorbing it, surfaces stay cooler under the sun. And for camouflage, that's a huge deal—it helps tanks or vehicles blend in with trees and plants when viewed through infrared detectors.
Cobalt aluminate is also being studied for use in gas sensors and catalysis. Its spinel structure is sensitive to certain volatile organic compounds, so it shows promise as a material for gas sensor elements. In catalysis research, it's being explored as a catalyst or catalyst support for cleaning up harmful components in car exhaust or industrial emissions. These studies are still in the lab phase.
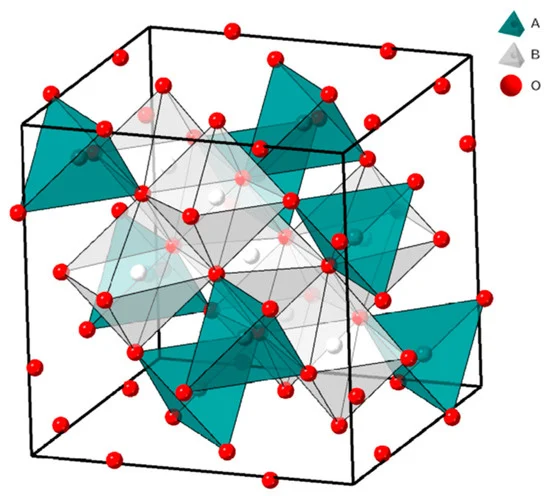
The crystal structure of AB2O4
Stanford Advanced Materials (SAM) offers high-quality Cobalt Aluminate Powder with competitive pricing. Inquiry to SAM
1. What plastics can cobalt aluminate pigment be used in? Are there any temperature limits?
It works with all sorts of plastics like PP, PE, PVC, PC, PA, and PPS. The spinel structure of cobalt aluminate is stable up to 300°C, so it can easily handle the processing temperatures of these plastics.
2. Will this pigment fade if used outdoors?
Cobalt aluminate has excellent weather resistance for an inorganic pigment. It doesn't absorb UV light, so it avoids the photochemical degradation that organic pigments often suffer from. Even after years of sun exposure, its color change is usually less than other blue pigments.
3. Is the cobalt blue pigment safe?
Yes, it is. The cobalt compounds may act as potential sensitizers. However, there is a potential for an allergic reaction in humans. When handling the powder, it is recommended that protective gloves and a dust mask be used to avoid inhalation of the powder.
4. What is the reason for the difference in the colors of the cobalt blue pigments I buy?
The color of the cobalt aluminate is quite sensitive to the conditions of manufacture. The calcination conditions and the particle size of the starting materials may all play a role in the final product. Some manufacturers may also dope it with other elements like zinc and lithium to improve the final product.
5. What is the test for good-quality cobalt blue?
In general, a good-quality cobalt blue will have a pure and vibrant color. A simple test is to heat the material at 300°C for 30 minutes and then cool it. A good-quality material will not change in color.

United States




